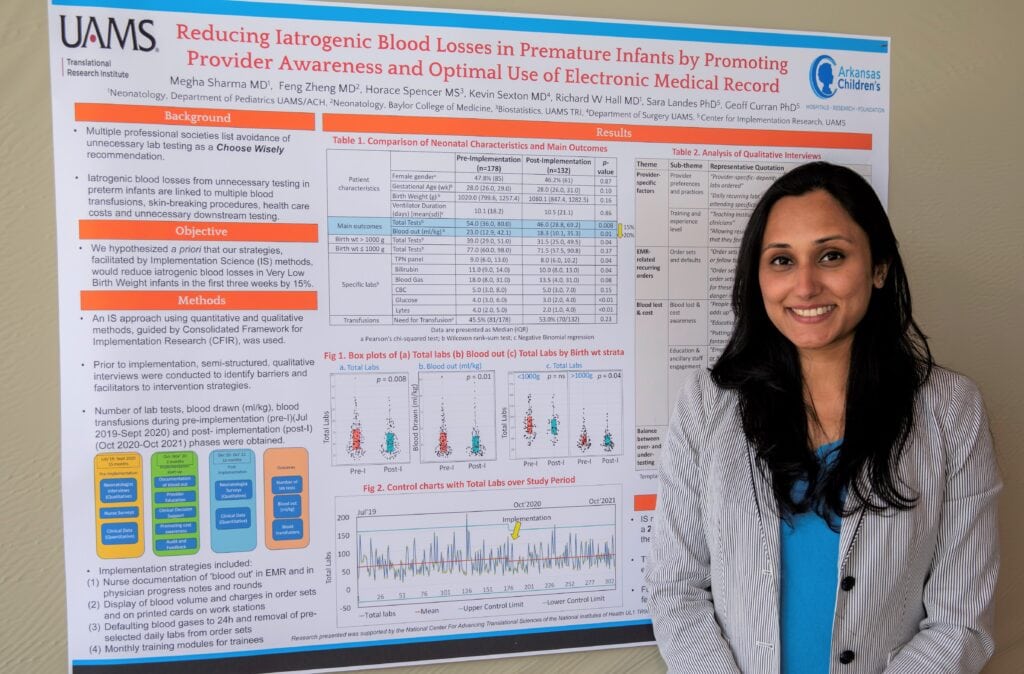
A successful effort at UAMS to reduce the amount of blood taken for lab tests from premature infants has been published in the journal Pediatrics.
The project was led by Megha Sharma, M.D., a neonatologist and associate professor in the College of Medicine Department of Pediatrics. Her work was conducted in the UAMS Neonatal Intensive Care Unit (NICU) over two years as part of the UAMS Translational Research Institute’s Implementation Science Scholars Program. Her paper, “Reducing Iatrogenic Blood Losses in Premature Infants,” was published in September.
Research has shown that blood loss from repetitive lab testing is a significant contributor to anemia in very low birth weight infants (less than 3.3 pounds). The blood lost from lab tests in the first few weeks of life often equals or exceeds the amount of an infant’s total blood volume, which is only 2-3 ounces. It can lead to a range of poor health outcomes.
“I was excited to see that we were able to achieve an 18% reduction in lab tests and blood loss,” Sharma said. “It has the potential over time to reduce the number of blood transfusions and the risk of anemia for our preterm babies.”
A key implementation strategy was revising automated patient care instructions in the electronic health record known as order sets that are used by NICU doctors.
“We had to take some of those defaults out of the computer-generated order sets so that our NICU physicians would be empowered to consider whether a blood test is actually needed,” Sharma said.
Helping physicians find the right balance of testing for such infants also involved resident education and an awareness campaign for all NICU care providers. Sharma used a travel-size 2-ounce bottle of hand sanitizer to demonstrate just how little blood the infants have available to give.
“An important aspect of this project was seeking qualitative input from physicians to thoroughly assess the NICU’s practices and culture so that we could tailor the interventions and deploy the most effective implementation strategies possible,” she said.
Resident physicians who rotate through the NICU monthly received laminated pocket cards stating the amount of blood needed for the 15 common lab tests in the NICU as well as the charges for those tests. This information also was provided in the electronic health record when entering lab orders.
Other implementation tactics included requiring documentation of the amount of blood drawn from each infant.
The project involved 354 infants and resulted in an 18.5% reduction in lab tests in the first three postnatal weeks, a 17% decrease in blood taken, and a $290,328 reduction in lab charges.
“There were fewer painful blood draw procedures and fewer invasive (central line) catheters placed as a result of this initiative,” Sharma said. The reduction in lab tests ordered also had no adverse health effects for the infants during the implementation science project.
In addition to her excitement about these results, Sharma said the implementation science project demonstrated how improvements can be made in other areas of a hospital.
“This work is generalizable,” she said. “Our methods can be adopted to reduce other unnecessary and wasteful practices, especially in an ICU setting. It involves modifying our way of thinking and embracing ‘less is more’ such that it is more respectful not only of patients but also of our economic and environmental resources.
The Implementation Science Scholars Program, led by Geoffrey Curran, Ph.D., made her project possible.
“I had identified a need for de-adopting a nonevidence-based practice, which is always a challenge. The Implementation Science program gave me a scientific and systematic way to approach it,” she said. “Dr. Curran was always present whenever I ran into barriers or needed help, and he and Dr. Sara Landes [Ph.D.] were instrumental in the success of this project. I am grateful for our biostatistician, the late Trey Spencer, for his incredible work in analyzing the effects of our interventions so we could disseminate our results to a wide community of physicians and researchers.”
Sharma said the knowledge gained in the Implementation Science program, which began in 2020, has informed her work as a student in the Clinical and Translational Science Master of Science program, supported by the Translational Research Institute, and as a current scholar in the institute’s K12 Mentored Research Career Development Program. The K12 program offers two years of translational research training, salary support and seed funding for a research project.
TRI is supported by the National Center for Advancing Translational Sciences at the National Institutes of Health, Clinical and Translational Science Award UM1 TR004909.
