Cite a Translational Research Institute CTSA award only when that specific award directly supported the work being reported. If the work was supported entirely by another source, or the Translational Research Institute (TRI) support was limited to routine services , do not cite the award in the publication.
Click here to view our updated citation manual.
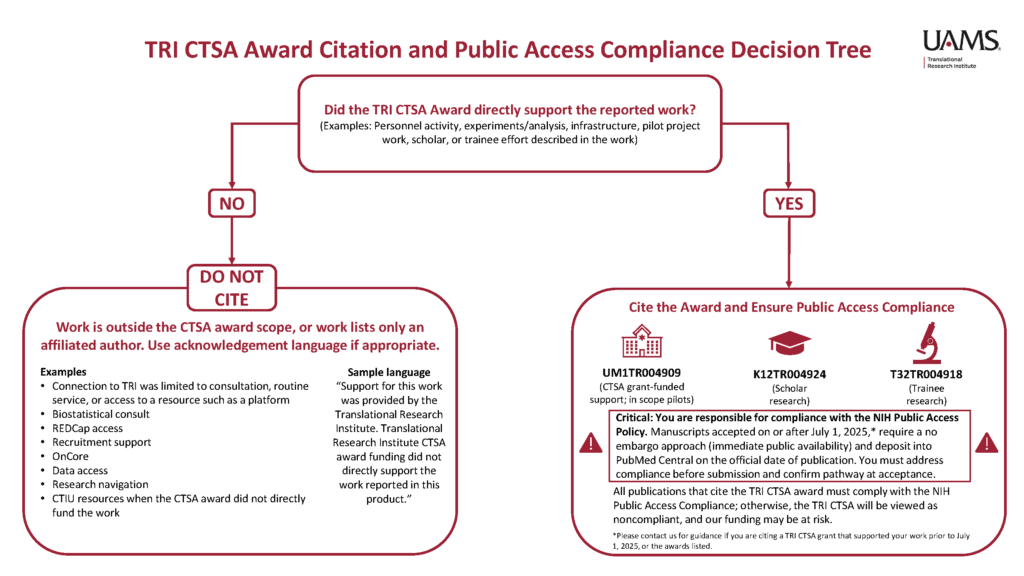
The NIH Public Access Policy applies to any Author Accepted Manuscript accepted for publication in a journal, on or after July 1, 2025.
This policy requires how we approach research publications citing our awards. It is critical that you pay close attention to these requirements, especially if you are the Primary Investigator (PI) or First Author on research papers you are submitting for publication. The landscape of NIH compliance has changed, and your oversight is the key to our collective success.
Citation Equals Compliance Responsibility
If you cite a TRI CTSA award in a journal article or other product, you are representing that the award directly supported the work. This triggers significant responsibilities:
- NIH Public Access Policy: You are responsible for ensuring the article is compliant with NIH rules.
- No Embargo: For articles accepted for publication on or after July 1, 2025, NIH requires immediate open access.
- Immediate Availability: The article must be available in PubMed Central on the Official Date of Publication.
- Author Oversight: Authors must confirm the compliance pathway before submission and monitor the deposit process until complete.
The Author’s Primary Responsibility
Compliance is the direct responsibility of the author — the individual who holds the contract with the research journal. Because only you have that formal relationship with the publisher, only you can ensure you are in full compliance with the NIH’s current Public Access Policy. While the TRI is here to provide guidance and support, the primary responsibility for compliance rests with you.
CRITICAL: Funding Risks of Non-Compliance
Why this matters: If our CTSA hub is not in compliance with the NIH Public Access Policy, it may delay or even stop our funding renewal at the beginning of the new grant year.
The Ripple Effect: If ANY publication that cites our CTSA grant number is found to be out of compliance, the entire CTSA hub is considered non-compliant. This jeopardizes the funding that supports our entire research community. We need you to be vigilant and help us retain our funding by ensuring every cited paper follows these rules.
The New Citation Standard
To ensure we remain in compliance and retain our funding, we are asking you to adopt a new mindset regarding how you credit the institute:
- Cite the grant number ONLY IF the award provided direct funding for the research being reported. By using the grant number (UM1 TR004909, K12 TR004924, and T32 TR004918), you are confirming that you will ensure the publication follows the NIH policy. This includes ensuring the paper is assigned a PubMed Central ID (PMCID) – which is different from a PMID – and is available immediately upon publication with no embargo.
- Acknowledge TRI if you used our resources, such as REDCap, Biostatistics, Informatics, or the Community Engagement Core, but did not receive direct funding. Acknowledgement without using the grant number allows us to track our impact without triggering the same NIH Public Access Policy requirements.
Example Citations and Acknowledgement Language
When TRI’s CTSA award Directly Supported the work (for example, personnel activity, experiments/analysis, infrastructure, pilot projects, scholar/trainee effort described in the product):
“Research reported in this publication was directly supported by the National Center for Advancing Translational Sciences of the National Institutes of Health under award number (insert appropriate grant number(s) here). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. This manuscript is the result of funding in whole or in part by the National Institutes of Health (NIH). It is subject to the NIH Public Access Policy. Through acceptance of this federal funding, NIH has been given a right to make this manuscript publicly available in PubMed Central upon the Official Date of Publication, as defined by NIH.”
When TRI provides resources or services to support the product (for example, use of REDCap, TriNetX, N3C, grant consultations, community engagement consultations, use of the equipment library, regulatory support):
“Support was provided by the Translational Research Institute. The Translational Research Institute’s CTSA funding did not directly support the work reported in this product.”
Which award numbers should I use?
When citing TRI-related awards, you should cite all award numbers for the period during which you have received TRI’s support. Support is determined by the date of award.
Multiyear awards should cite award numbers that correspond with both the initial notice of award date and any subsequent funding years. For example, an awardee who received year one of funding in 2023 and year two in 2024 would need to cite both UL1 TR003107 and UM1 TR004909.
Use the tables below to identify which award number(s) you should cite.
Support received between 2024-2029:
| Sponsor | Start Date | End Date | Award ID |
| NCATS | 7/1/2024 | 6/30/2029 | K12 TR004924 |
| NCATS | 7/1/2024 | 6/30/2029 | T32 TR004918 |
Support received between 2024-2031:
| Sponsor | Start Date | End Date | Award ID |
| NCATS | 7/1/2024 | 6/30/2031 | UM1 TR004909 |
Support received between 2019-2024:
| Sponsor | Start Date | End Date | Award ID |
| NCATS | 7/1/2019 | 6/30/2024 | UL1 TR003107 |
| NCATS | 7/1/2019 | 6/30/2024 | KL2 TR003108 |
| NCATS | 7/1/2019 | 6/30/2024 | TL1 TR003109 |
Support received between 2009-2019:
| Sponsor | Start Date | End Date | Award ID |
| NCATS | 9/26/2017 | 6/30/2019 | U54 TR001629 |
| NCATS | 4/1/2012 | 3/31/2016 | KL2 TR000063 |
| NCATS | 4/1/2012 | 3/31/2016 | UL1 TR000039 |
| NCRR | 7/14/2009 | 3/31/2012 | UL1 RR029884 |
| NCRR | 7/14/2009 | 3/31/2012 | KL2 RR029883 |
Still have questions? Please contact Mtonya Hunter (HunterLewisMtonya@uams.edu).
Helpful Links for NIH Public Access Policy
New NIH Public Access Pages to Support the 2024 NIH Public Access Policy
2024 NIH Public Access Policy Notice Number: NOT-OD-25-047
Supplemental Guidance to the 2024 NIH Public Access Policy: Publication Costs. Notice NOT-OD-25-048
Special thanks to our colleagues at the C. Kenneth and Dianne Wright Center for Clinical and Translational Research CTSA for their collaboration in developing this guidance for our hub.
